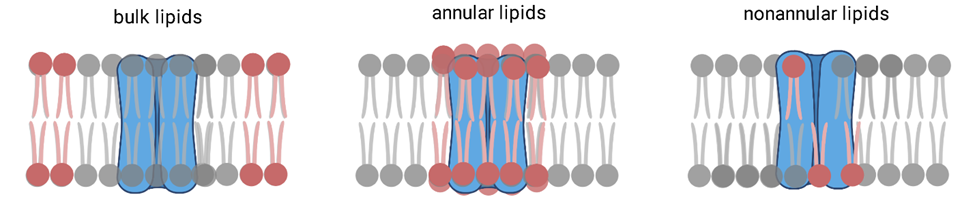
Over 30% of the human proteome consists of membrane proteins. These proteins are critical in many biological processes and are implicated in a large number of diseases. Previously, it was thought that lipids act only as a solvent for membrane proteins, but now it is widely recognized that lipids play a vital role. Membrane lipids interact with proteins in various ways, ranging from stabilizing conformations, embedding into the membrane, or activating some function. Protein-lipid interactions include specific lipid binding to the protein or non-specific interactions by altering the properties of the membrane in which the protein resides. It is challenging to characterize these interactions separately because the likelihood of random interactions between the protein and lipids is prevalent due to the high ratio of lipids to protein in the plasma membrane, being around 75:1. In the Meilerl Laboratory, we leverage structural data in conjunction with evolutionary computations to detect likely-specific interaction sites of lipids on the surface of proteins. Computational protein-lipid design has the potential to provide a general, complementary approach to engineering lipid recognition into proteins.
References:
[1]
Brennica Marlow, Georg Kuenze, Bian Li, Charles R. Sanders, Jens Meiler,
Structural determinants of cholesterol recognition in helical integral membrane proteins,
Biophysical Journal, Volume 120, Issue 9, 2021 https://doi.org/10.1016/j.bpj.2021.02.028.
