HoxA11 is a transcription factor that plays a critical role in embryonic development and is required for female fertility. Like all Hox proteins, HoxA11 contains a highly conserved homeodomain that binds DNA. However, the rest of the protein shows low levels of secondary structure, large stretches of disordered regions, and no easily detectable similarity to any fold in the PDB. De novo structure prediction using ROSETTA reveals a region outside of the homeodomain that folds independently into a small helical bundle similar to those seen in different DNA binding domains. We are pursuing structural characterization of HoxA11 to test this hypothesis and to elucidate the molecular basis of Hox-dependent transcriptional activation mechanisms. Since this region of the protein also contains sites that are believed to be important in the evolution of pregnancy in mammals, our results may provide insight into how novel developmental pathways evolve.
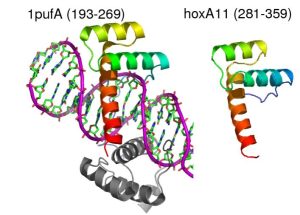
Comparative model of the HoxA11 homeodomain based on the crystal structure of HoxA9 (1pufA)
Alumni Project Members: Laura Mizoue, Caitlin Heuberger, Joel Harp, Soumya Ganguly, Heather Darling
